1. Introduction
In the age of rapid medical advancement , electronic technology has become an indispensable part of patient diagnosis, monitoring, and treatment. From wearable health devices that track vital signs to complex surgical robots assisting in intricate procedures, the demand for reliable and highly specialized electronics continues to grow. Medical PCBs are the silent workhorses, providing the necessary electrical connections and support for electronic components, ensuring devices function exactly as intended, every single time.
The importance of these specialized PCBs cannot be overstated. A failure in a medical device can have severe, even life-threatening, consequences. This necessitates a level of precision, quality, and regulatory compliance that far exceeds that of consumer or even industrial electronics. It’s a field where innovation constantly pushes boundaries while remaining anchored by an unwavering commitment to safety and reliability.
Key Takeaways
- Medical PCBs are highly specialized circuit boards essential for the functionality and safety of healthcare devices.
- They differ from standard PCBs due to requirements for extreme reliability, precision, safety, and sometimes biocompatible materials.
- Key regulatory standards include IPC Class 3 , FDA requirements, and ISO 13485.
- Applications span diagnostic equipment , therapeutic devices, monitoring systems, and implantable devices , often demanding miniaturization and robust signal integrity.
- Choosing a manufacturer with proven expertise, certifications, and strong quality control is crucial for medical device success.
2. What Exactly is a Medical PCB?
A Medical PCB is a printed circuit board specifically designed, manufactured, and assembled for use in medical devices. Unlike general-purpose PCBs, Medical PCBs must adhere to extremely strict health and safety standards, performance requirements, and regulatory guidelines. They are tailored to the unique demands of healthcare environments, which often involve sterile conditions, high reliability, specific signal integrity needs, and sometimes even direct contact with the human body.
These PCBs are fundamental to the functionality of medical devices, supporting everything from data processing in a patient monitor to precise motor control in surgical robots. Their design and fabrication process is a specialized undertaking that requires in-depth knowledge of not only electronics but also biomedical engineering principles and regulatory frameworks.
3. Why Medical PCBs Are Different: Key Characteristics
The distinguishing factors of Medical PCBs stem from the critical nature of their applications. They are engineered for scenarios where failure is not an option. Here are the key characteristics that set them apart:
- Uncompromising Reliability: Medical devices often operate continuously and must maintain peak performance over long periods. Whether it’s an implantable device or a diagnostic machine, the PCB must be incredibly robust and fault-tolerant.
- High Precision and Accuracy: Many medical applications, such as imaging systems or therapeutic devices, require extreme precision. The PCB must support stable power delivery, minimal signal noise, and exact component placement to ensure accurate data acquisition and operation.
- Safety and Biocompatibility: For devices that come into contact with patients, or are even implanted, the materials used must be non-toxic and biocompatible, meaning they will not cause adverse reactions in the human body.
- Miniaturization: There’s a constant drive for smaller, less intrusive medical devices, especially in wearables and implantables . This leads to the frequent use of High-Density Interconnect (HDI) PCBs, flexible PCBs, and rigid-flex designs to maximize functionality in minimal space.
- Sterilization Compatibility: PCBs in devices used in sterile environments must withstand various sterilization methods (e.g., autoclaving, ETO, gamma radiation) without degradation of performance or material integrity.
- Long Lifespan and Traceability: Medical devices typically have a long service life, and every component, including the PCB, must be traceable back to its origin and manufacturing batch for quality control and recall purposes.
4. Specialized Materials for Medical PCB Manufacturing

The choice of materials is crucial for Medical PCBs, impacting everything from biocompatibility and thermal management to signal integrity and sterilization resistance. Common materials include:
- Substrates: High-performance laminates like FR- 4 (with enhanced properties), Polyimide, or BT Epoxy are often used for their excellent electrical properties, thermal stability, and mechanical strength. For flexible applications, materials like Kapton are essential.
- Biocompatible Materials: When direct patient contact or implantation is involved, materials must be rigorously tested for biocompatibility. This applies not only to the substrate but also to solders, conformal coatings, and inks.
- Low Loss Materials: For high -frequency medical imaging or diagnostic equipment, materials with low dielectric loss are crucial to maintain signal integrity and minimize attenuation.
- Thermal Management Materials: Devices that generate significant heat (e.g., MRI imaging systems) require PCBs with specialized thermal properties, often incorporating heavy copper layers or thermal vias to dissipate heat effectively.
- Conformal Coatings: These protective layers safeguard the PCB from moisture, chemicals, and mechanical stress, extending device longevity, especially in harsh environments.
5. Navigating Regulatory Standards and Certifications
The regulatory landscape for Medical PCBs is complex and rigorous. Adherence to these standards is non-negotiable for manufacturers aiming to supply the medical industry. Key regulatory bodies and standards include:
5.1. IPC Class 3
For medical devices, the industry standard is almost always IPC Class 3, as specified by the IPC-A-610 standard. This class designates the highest level of reliability for electronic products where continuous performance is critical. It mandates extremely stringent requirements for solder joint quality, component placement, material selection, and overall manufacturing processes , ensuring the highest possible reliability for critical applications where downtime is unacceptable.
| IPC Class | Description | Typical Applications |
| Class 1 | General electronic products (least stringent) | Consumer electronics, toys |
| Class 2 | Dedicated service electronic products (longer life expected) | Industrial equipment, telecommunications |
| Class 3 | High-reliability electronic products (continuous performance, critical downtime) | Medical devices, aerospace, military |
5.2. FDA Requirements
In the United States, the Food and Drug Administration (FDA) regulates medical devices. Manufacturers of Medical PCBs and assemblies must comply with FDA’ s Quality System Regulation (QSR) 21 CFR Part 820. This regulation mandates a robust quality management system (QMS) covering design, manufacturing, packaging, labeling, storage, installation, and servicing of medical devices. While the FDA doesn’t directly certify PCBs, compliance is essential for the final device’s approval.
5.3. ISO 13485
ISO 13485 is an internationally recognized standard for quality management systems specific to medical devices. It outlines comprehensive requirements for manufacturers to ensure consistency, reliability, and safety throughout the product lifecycle. A medical PCB manufacturer that is ISO 13485 certified demonstrates a commitment to meeting these stringent quality and regulatory expectations, which is a key factor when choosing a supplier.
6. Diverse Applications of Medical PCBs in Healthcare
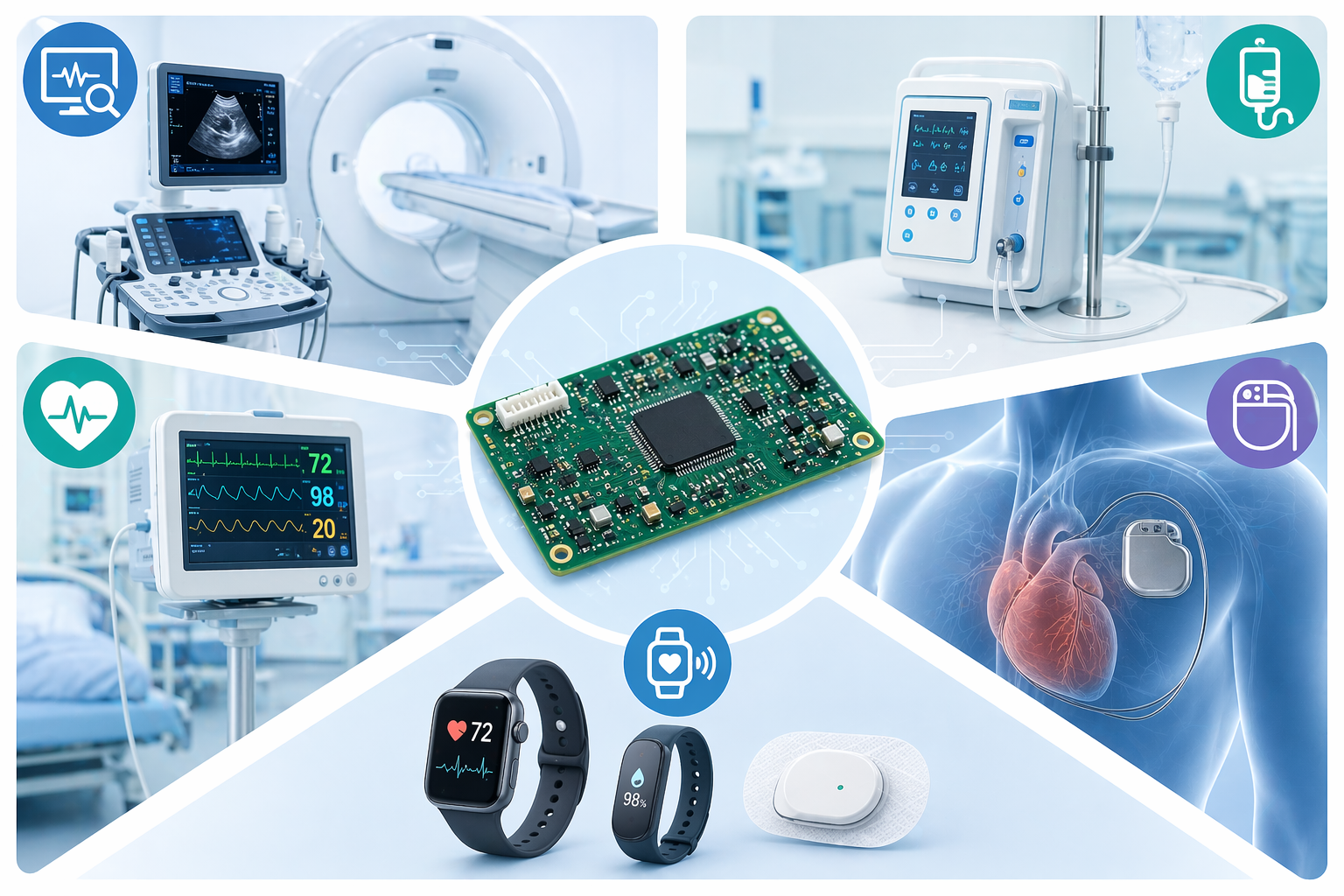
Medical PCBs are integral to a vast array of devices across diagnostics, therapeutics, monitoring, and even implantable solutions. Here are some key application areas:
- Diagnostic Equipment: These include advanced imaging systems such as MRI imaging systems, CT scanners, X-ray machines, and ultrasound devices. Also, blood glucose monitors, ECG/EKG machines, and various laboratory diagnostic equipment rely heavily on precise PCB functionality.
- Therapeutic Devices: This category encompasses life-saving devices like pacemakers, defibrillators, nerve stimulators, and drug delivery systems. Complex devices like surgical robots and laser surgical tools also depend on high-reliability Medical PCBs for precise control and operation.
- Monitoring Systems: From patient monitors that track vital signs in intensive care units to sleep apnea monitors and neonatal monitoring systems, Medical PCBs ensure continuous, accurate data collection and display.
- Implant able Devices: These are perhaps the most critical applications, including pacemakers, cochlear implants, neurostimulators, and internal defibrillators. These PCBs must be incredibly small, durable, and biocompatible, designed for decades of reliable operation within the human body.
- Wearable Health Devices: The rise of wearable health devices and remote patient monitoring has created a demand for flexible, miniaturized, and low-power PCBs. These devices include smartwatches, fitness trackers, and continuous glucose monitors.
7. Design and Manufacturing Considerations
Creating Medical PCBs presents unique challenges beyond standard PCB design:
- Miniaturization and Density: The push for smaller, lighter devices often requires High-Density Interconnect (HDI) designs, microvias, and fine-pitch components. This demands advanced manufacturing techniques and rigorous quality control.
- Signal Integrity: Ensuring clean, accurate signal transmission is vital, especially for sensitive diagnostic equipment. This involves careful impedance control, precise routing, and robust grounding schemes.
- Thermal Management: Heat dissipation is a significant concern, particularly in compact devices or those handling high power. Effective thermal management strategies are integrated into the PCB design, such as heavy copper layers or thermal vias.
- Cleanliness and Sterility: Manufacturing environments must be exceptionally clean to prevent contamination. Post-fabric ation, PCBs may need to withstand sterilization processes without compromising performance.
- Testing and Validation: Medical PCBs undergo extensive testing, including electrical testing (e.g., flying probe, in-circuit test), functional testing, environmental testing (e.g., temperature, humidity cycling), and burn-in testing to ensure long-term reliability.
8. Choosing the Right Medical PCB Manufacturer
Given the high stakes involved, selecting a reputable and experienced Medical PCB manufacturer is paramount. Key factors to consider include:
- Certifications: Ensure the manufacturer holds relevant certifications like ISO 134 85 and adheres to FDA QSR.
- IPC Class 3 Capability: Verify their proven ability to consistently produce boards to the stringent IPC Class 3 standards.
- Experience: Look for a track record in manufacturing PCBs for medical applications, demonstrating an understanding of the specific demands of the industry.
- Quality Control: A robust quality assurance system, including thorough inspection, testing, and traceability protocols, is essential.
- Engineering Support: A manufacturer who can provide design-for-manufacturability (DFM) feedback and collaborate on optimizing designs for reliability and cost is invaluable.
- Material Expertise: They should have access to and experience with the specialized materials required for medical devices, including biocompatible options.
Partnering with an expert manufacturer is not just about building a PCB; it’s about entrusting them with a critical component that will safeguard lives and enhance healthcare outcomes.
FAQ
Question 1: What is the most critical standard for Medical PCBs?
Answer: While FDA and ISO 13485 are crucial for the overall quality management system, IPC Class 3 is the most critical technical standard for the manufacturing and assembly quality of the PCB itself, ensuring the highest level of reliability required for medical devices.
Question 2: Are all Medical PCBs rigid?
Answer: No, many Medical PCBs are flexible or rigid-flex designs, especially for implantable devices, wearables, or applications requiring compact, irregularly shaped electronics that can bend or conform to specific areas.
Question 3: How do Medical PCBs handle sterilization?
Answer: Medical PCBs intended for sterile environments are designed with materials and finishes that can withstand various sterilization methods, such as autoclaving (steam and high heat), ethylene oxide (ETO) gas, or gamma radiation, without degrading their performance or structural integrity.
Summary
Medical PCBs are far more than just electronic components; they are the unseen heroes enabling the precision, reliability, and innovation that define modern healthcare. Their design and manufacturing demand an unparalleled commitment to quality, adherence to strict regulatory standards like IPC Class 3, FDA, and ISO 13485, and the use of specialized, often biocompatible materials. From diagnostic equipment and therapeutic devices to implantable devices and wearable health devices, these specialized circuit boards ensure that medical technology functions flawlessly, directly contributing to improved patient outcomes and advancements in medicine.
 Isaiminia World Breaking News & Top Stories
Isaiminia World Breaking News & Top Stories




